The world is buoyed by the hope of a COVID-19 vaccine to help us move past the pandemic. In this post, we discuss CoronaVac, a front-runner in the COVID-19 vaccine race.
Internationally, scientists have been working at an impressive pace to learn more about COVID-19 and develop novel vaccines and therapeutics. Worldwide, we all hope to have an answer to the question, “When will the pandemic end?” It is becoming clear that a COVID-19 vaccine will need to be a crucial tool to combat COVID-19.
The race to a vaccine got a good start in January 2020, when China published the genetic sequence for SARS-CoV-2, the virus that causes COVID-19. Since then, dozens of laboratories around the world are now the battlegrounds in the race to develop a COVID-19 vaccine.
China is a leader in the race to a vaccine, with the most number of COVID-19 vaccines currently in human trials. By June 2020, six Chinese companies had introduced COVID-19 vaccine candidates in human trials.
Sinovac’s CoronaVac Vaccine
The most promising of the Chinese COVID-19 vaccines currently in human trials is CoronaVac. CoronaVac is under development by a Beijing-based pharmaceutical company called Sinovac Biotech. The company has developed human vaccines for conditions such as hepatitis A and B, and has also led efforts to develop vaccines for avian flu and swine flu.
Currently in Phase 3 trials in Brazil, Sinovac’s CoronaVac vaccine is a frontrunner in the race to develop a COVID-19 vaccine. Sinovac’s ambitious goal is to have CoronaVac ready for the market by late 2020, but, of course, approval for patient use hinges on successful completion of the trials.
CoronaVac, Moderna, Oxford: Three Different Vaccines, One Goal
Others leading the way in this international race to a vaccine for the novel coronavirus are the United Kingdom’s Oxford/AstraZeneca vaccine and the United States’ Moderna vaccine.
What’s interesting to note is the top three vaccines are all different types. The Oxford/AstraZeneca vaccine uses viral vectors, and the Moderna vaccine uses mRNA. By contrast, CoronaVac uses a modified version of the SARS-CoV-2 virus itself.
Furthermore, Oxford and Moderna have something in common with each other that they do not have in common with CoronaVac. The methods used by both Oxford and Moderna rely on the latest tools of modern biology to foster immunity to a small part of the SARS-CoV-2 virus that causes COVID-19. In both the Moderna and Oxford vaccines, the human body develops immunity to a small part of the SARS-CoV-2 virus — the spike protein, a part of the SARS-CoV-2 molecule which mediates viral infection.
By contrast, CoronaVac uses the whole, inactivated virus — not just the spike protein — as the basis of the vaccine. CoronaVac uses an age-old method of growing the SARS-CoV-2 virus in bulk, and then inactivating it using chemicals. The method used by CoronaVac has been used consistently throughout history, with the polio vaccine being the most notable of them all.
What is an Inactivated Vaccine?
Inactivated vaccines are made by inactivating, or killing, the pathogens (that is, bacteria or viruses) during the process of vaccine development. Heat or chemicals such as formaldehyde are used to inactivate viruses for use in vaccines. The main benefit of inactivated viruses — and why they are used in vaccines — is that they exhibit the whole virus to the immune system, and because they are killed, cannot cause disease.
The inactivated vaccine is the first type of vaccine that was ever invented. The history of vaccines, and of inactivated vaccines in particular, goes back to the late 18th century, when an English doctor named Edward Jenner, who is widely known as the father of immunology, developed a vaccine for smallpox. Jenner’s smallpox vaccine helped eliminate a very serious disease that “devastated mankind for centuries,” as Dr. Stefan Riedel writes. Thanks to this vaccine, smallpox was eradicated in 1980.

How Do Inactivated Vaccines Work?
Inactivated vaccines work because they cannot cause disease — they have been treated with heat or chemicals to render them unable to actively cause disease in the body. However, once an inactivated vaccine is injected into the body, the immune cells in our bodies will recognize the inactivated pathogens (in CoronaVac’s case, the inactivated SARS-CoV-2 virus) as “foreign.” This then induces an immune response against the already inactivated pathogen.
The immune response to the inactivated pathogen ensures that the memory cells of our immune system know the “cheat codes” for the next time it is exposed to the pathogen we vaccinate against. Using these “cheat codes,” our bodies are guaranteed to win the fight against the pathogen for which it receives a vaccine that we may naturally be exposed to at a later time.
Inactivating the SARS-CoV-2 virus
As we’ve just discussed, vaccines train our immune systems to fight off diseases caused by pathogens like bacteria and viruses. CoronaVac, an inactivated vaccine, uses a killed version of the SARS-CoV-2 virus. The infectious agent (in this case, the SARS-CoV-2 virus) is grown in the laboratory, and then inactivated or rendered uninfectious using chemicals.
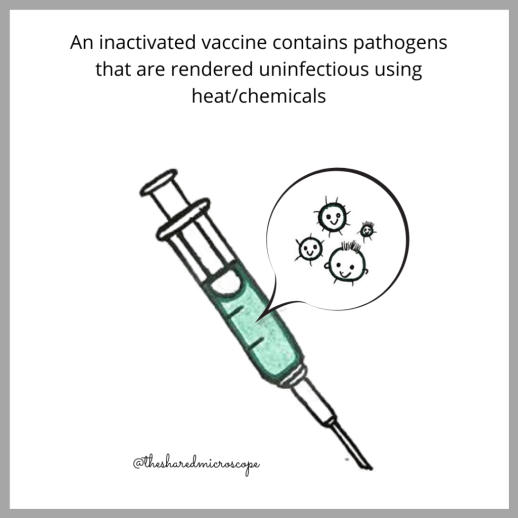
Since the virus is inactivated, it can no longer replicate and cause infection or even replicate when injected into a human. For this reason, this type of vaccine is also safe for use in an immunodeficient person.
Once injected with CoronaVac’s vaccine, our immune cells will be able to detect these inactivated free floating SARS-CoV-2 viral particles. Following the process explained above, the vaccine will help our bodies identify the SARS-CoV-2 viral particles as “foreign,” and thereby eliminate them when naturally exposed at a later date.
Since the viruses in this vaccine are whole inactivated viruses, the vaccine may not be capable of evoking a very robust immune response – the vaccine may therefore require the recipient to take booster shots to ensure a robust immune response. It should also be noted that because the viruses are inactivated, a higher dose is usually required to immunize someone.
Experimental Studies Support CoronaVac’s Utility
To develop CoronaVac, multiple strains of the SARS-CoV-2 virus, isolated from 11 different hospitalized patients, were grown and then chemically inactivated. Animal trials were conducted using the inactivated viral particles.
Mice were first injected with differing doses of the experimental vaccine at days 0 and 7. This was done to check for inflammation and adverse effects. No inflammation or adverse effects were observed in the mice. Similar results were also observed in rats.
After the initial study to check immunogenicity in mice, the vaccine was trialled in rhesus monkeys. The vaccine produced neutralising antibodies in rhesus monkeys. The CoronaVac vaccine was able to successfully pass animal trials. A total of eight rhesus macaques were injected with two different doses of the experimental COVID-19 vaccine being developed by Sinovac. The results showed a strong protective response against the SARS-CoV-2 virus.
The monkeys were each injected with the experimental vaccine. The monkeys each received three immunisations using either the 3 microgram dose or the 6 microgram dose, respectively.
Three weeks later, the SARS-CoV-2 virus, which causes COVID-19, was introduced to the monkeys’ lungs through tubes down their windpipes.
The results showed that monkeys that received the lower (3 microgram) dose of the experimental CoronaVac vaccine showed signs of controlling the infection. For the monkeys that received the higher (6 microgram) dose of the vaccine, researchers were unable to detect any virus particles in the throats or the lungs of the monkeys. Monkeys receiving the 6 microgram doses showed complete protection when exposed to the SARS-CoV-2 virus. Monkeys that were not given either doses of the experimental vaccine had excessive levels of viral RNA in their throats and lungs, and also developed severe pneumonia.
Due to the results of the above experiments, it is believed that the vaccine will have protective properties when injected in humans. What is also of relevance is that the antibodies produced via the experimental vaccine developed by Sinovac appeared to be effective against at least ten different mutational forms of the SARS-CoV-2 virus.
This study also addresses a concern common to all vaccines so far – the concern of pulmonary immunopathology. In other words, a number of concerns have been raised with regards to the possibility that the vaccines accentuate COVID-19 pathogenesis by eliciting a ‘cytokine storm’, via specific immune cells called the T cells. T cell responses elicited by any SARS-CoV-2 vaccine therefore needs to be controlled to ensure that T cell responses are managed so as to not cause further COVID-19 infection. The researchers therefore kept a close eye on the T cell response, and observed no immunopathology in the vaccinated macaques whatsoever.
CoronaVac Clinical Trials
Following success in preclinical research done on animal models, CoronaVac commenced clinical trials in Spring of 2020. The CoronaVac vaccine is one of several Chinese companies that have gotten approval to continue on to human trials. If human trials are successful, Sinovac Biotech is on track to make 100 million vaccine doses per year.
Sinovac started human trials in April to test the safety of the CoronaVac vaccine. In June 2020, Sinovac announced the results of their Phase I/II clinical trials. In these trials, 743 healthy volunteers aged 18 to 59 years old received the experimental CoronaVac vaccine. Of the 743, 143 volunteers were in Phase I studies and 600 volunteers in Phase II. Taken together, the results of the trials showed that there were no severe adverse events reported in either Phase I or in Phase II clinical trials. Sinovac also announced that the results of their Phase II trials showed that the vaccine was able to produce neutralizing antibodies 14 days after vaccination. The vaccine therefore looks promising as a candidate that will help us control the COVID-19 pandemic.
Since Sinovac completed the Phase I/II studies, they could go ahead into Phase III trials. One of the biggest obstacles that the company was facing, for the Phase III trials, was the lack of COVID-19 transmission in China. For this reason, Sinovac applied for approval to carry out their trials internationally.
Sinovac Biotech is now collaborating with Instituto Butantan in Brazil to conduct Phase III clinical trials. They have as of July 03, 2020 received approval for phase 3 trials in Brazil. The trial in Brazil is pivotal to support the licensure of this vaccine. Nearly 9,000 healthcare professionals working frontline to manage the COVID-19 crisis are set to receive this experimental vaccine developed by Sinovac Biotech.
Currently, the hope is that we hear more about the safety and efficacy of the CoronaVac vaccine and the other vaccines in Phase III clinical trials as early as Fall 2020. It should be noted though, that a vaccine is not the answer to ending the pandemic. What we need is a multifaceted approach to ensure a vaccine is developed and delivered to the majority of the population worldwide, and until we can do that, social distancing rules must be followed and testing and tracing must continue.
This post was originally written for Fancy Comma, LLC. Check out their Twitter, Facebook, LinkedIn, or Instagram for more information, or visit their blog for more insights on effective science writing.
Related posts:
 Covid-19: Let’s Get Handsy!
Covid-19: Let’s Get Handsy!
 [Newsletter] Round-Up of COVID-19 Vaccine Updates: Week Commencing September 28, 2020
[Newsletter] Round-Up of COVID-19 Vaccine Updates: Week Commencing September 28, 2020
 [Newsletter] Round-Up of COVID-19 Vaccine Updates: Week Commencing November 09, 2020
[Newsletter] Round-Up of COVID-19 Vaccine Updates: Week Commencing November 09, 2020
 [Newsletter] Round-Up of COVID-19 Vaccine Updates: Week Commencing November 16, 2020
[Newsletter] Round-Up of COVID-19 Vaccine Updates: Week Commencing November 16, 2020
 [Newsletter] Round-Up of COVID-19 Vaccine Updates: Week Commencing January 18, 2021
[Newsletter] Round-Up of COVID-19 Vaccine Updates: Week Commencing January 18, 2021
- Written by: admin
- Posted on: July 22, 2020
- Category: COVID-19

0 comments
The View from the Armchair
July 23, 2020 at 12:49 amReblogged this on The View from the Armchair and commented:
Interesting. First I’ve read of this.
Shane
July 23, 2020 at 7:45 amThat’s a crisp and precisely written piece. Its refreshingly relevant and informative. Kudos.
Nidhi
July 23, 2020 at 12:42 pmThank you! We aim to write about the latest in science for the lay audience, especially in the midst of a pandemic! Scientifically correct information has never been as important as it is today!
Rebbecca Marich
July 27, 2020 at 7:46 amWONDERFUL Post.thanks for share..more wait .. …
real credit card that works
July 30, 2020 at 3:10 pmWonderful beat ! I would like to apprentice while you amend your website, how could i subscribe for a blog site?The account helped me a acceptable deal. I had been tiny bit acquainted of this your broadcast offered bright clear idea
Novavax: A SARS-CoV-2 “Protein Factory” to Beat COVID-19 – The Shared Microscope
August 3, 2020 at 10:47 am[…] CoronaVac, developed by Sinovac Biotech, which is an inactivated virus […]
Vaccine Development: A Chronology – The Shared Microscope
August 3, 2020 at 5:30 pm[…] posts. We have previously written posts on the Moderna vaccine, the Oxford/AstraZeneca vaccine, the CoronaVac developed by Sinovac, and the vaccine developed by […]
Mira Crapser
August 4, 2020 at 9:54 amThanks for your marvelous posting! I certainly enjoyed reading it, you might be a great author.I will remember to bookmark your blog and will come back sometime soon. I want to encourage that you continue your great work, have a nice holiday weekend!
Trey Pedri
August 7, 2020 at 2:00 pmMy husband and i got thrilled when Emmanuel managed to deal with his basic research via the precious recommendations he received through your blog. It’s not at all simplistic just to possibly be offering concepts which many people have been making money from. So we keep in mind we need the website owner to appreciate for that. Most of the explanations you’ve made, the straightforward website menu, the relationships you will help to engender – it’s got most great, and it is assisting our son in addition to the family do think that idea is awesome, which is certainly particularly important. Thank you for all the pieces!
Why the Anti-Vax Rhetoric Prevails – The Shared Microscope
September 1, 2020 at 6:59 pm[…] vaccines (the Moderna vaccine, Oxford University/AstraZeneca Vaccine, the vaccine in development by Sinovac, and the Novavax vaccine) in development […]
Ka Cohee
September 8, 2020 at 9:17 pmHello my friend! I want to say that this article is awesome, nice written and include approximately all important infos. I would like to see more posts like this.
Marlo Bozarth
September 11, 2020 at 12:03 amHi, I enjoy your blog and I enjoyed this article a whole lot. . How may I read more?
[Newsletter] Round-Up of COVID-19 Vaccine Updates: Week Commencing September 07, 2020 – The Shared Microscope
September 13, 2020 at 1:23 am[…] CoronaVac: A COVID-19 Vaccine Made From Inactivated SARS-CoV-2 Virus […]
Roselee Karren
September 19, 2020 at 2:43 pmIt is really a great and useful piece of information. I’m glad that you shared this useful info with us. Please keep us up to date like this. Thanks for sharing.
[Newsletter] Round-Up of COVID-19 Vaccine Updates: Week Commencing September 14, 2020 – The Shared Microscope
September 20, 2020 at 6:45 pm[…] How Does Sinovac’s CoronaVac, A COVID-19 Vaccine Made From Inactivated SARS-CoV-2 Virus, Work? […]
Pfizer and BioNTech’s mRNA Vaccine Against COVID-19: How Does It Work? – The Shared Microscope
September 23, 2020 at 2:41 pm[…] vaccine, the Oxford University/AstraZeneca vaccine, the vaccines in development by Novavax and Sinovac, is another frontrunner in this race against the […]
nigerianewspaperonline
October 2, 2020 at 5:13 amI really liked your blog.Really thank you! Awesome.
[Newsletter] Round-Up of COVID-19 Vaccine Updates: Week Commencing December 07, 2020 – The Shared Microscope
December 14, 2020 at 8:37 pm[…] Sinovac’s CoronaVac […]
[Newsletter] Round-Up of COVID-19 Vaccine Updates: Week Commencing November 30, 2020 – The Shared Microscope
December 14, 2020 at 8:39 pm[…] Sinovac’s CoronaVac […]
[Newsletter] Round-Up of COVID-19 Vaccine Updates: Week Commencing November 23, 2020 – The Shared Microscope
December 14, 2020 at 8:41 pm[…] Sinovac’s CoronaVac […]
[Newsletter] Round-Up of COVID-19 Vaccine Updates: Week Commencing December 14, 2020 – The Shared Microscope
December 21, 2020 at 9:08 pm[…] Sinovac’s CoronaVac […]
[Newsletter] Round-Up of COVID-19 Vaccine Updates: Week Commencing December 21, 2020 – The Shared Microscope
December 28, 2020 at 6:04 pm[…] Sinovac’s CoronaVac […]
[Newsletter] Round-Up of COVID-19 Vaccine Updates: Week Commencing December 28, 2020 – The Shared Microscope
January 4, 2021 at 7:49 pm[…] Sinovac’s CoronaVac […]
[Newsletter] Round-Up of COVID-19 Vaccine Updates: Week Commencing January 04, 2020 – The Shared Microscope
January 11, 2021 at 6:55 pm[…] Sinovac’s CoronaVac […]
[Newsletter] Round-Up of COVID-19 Vaccine Updates: Week Commencing January 11, 2021 – The Shared Microscope
January 18, 2021 at 4:22 pm[…] Sinovac’s CoronaVac […]
[Newsletter] Round-Up of COVID-19 Vaccine Updates: Week Commencing January 18, 2021 – The Shared Microscope
January 25, 2021 at 8:01 pm[…] Sinovac’s CoronaVac […]
[Newsletter] Round-Up of COVID-19 Vaccine Updates: Week Commencing January 25, 2021 – The Shared Microscope
February 1, 2021 at 5:48 pm[…] Sinovac’s CoronaVac […]
[Newsletter] Round-Up of COVID-19 Vaccine Updates: Week Commencing February 01, 2021 – The Shared Microscope
February 8, 2021 at 8:57 pm[…] Sinovac’s CoronaVac […]
[Newsletter] Round-Up of COVID-19 Vaccine Updates: Week Commencing February 08, 2021 – The Shared Microscope
February 15, 2021 at 7:32 pm[…] Sinovac’s CoronaVac […]