With the world slowly showing signs of returning to normalcy, all eyes are peeled for promising news about the COVID-19 vaccines. In this post, we discuss Novavax’s NVX-CoV2373, another vaccine in the COVID-19 vaccine race.
Scientists around the globe are carrying out groundbreaking research to produce a safe and effective vaccine by next year — a great feat considering the process to develop and release a vaccine into the market can typically take a couple of years. With more than 165 vaccines being developed against the novel coronavirus, of which at least 31 are in human trials, we are still a while away from bringing the pandemic under control.
In previous articles, we have covered a number of the frontrunners, including:
- The Oxford/AstraZeneca vaccine based on viral vectors
- The Moderna vaccine which uses RNA
- The CoronaVac, developed by Sinovac Biotech, which is an inactivated virus vaccine
The current post will introduce you to NVX-CoV2373, the vaccine under development by Novavax. This vaccine candidate relies on a protein subunit to establish immunity.
Read on to learn more about Novavax and the science behind this COVID-19 vaccine.
What is a Subunit Vaccine?
Simply put, a subunit vaccine uses a single component of a pathogen (that is, a bacterium or virus) to establish immunity. In other words, the vaccine is made from a single part of the pathogen — a subunit — rather than the whole pathogen itself.
In the image below, our hypothetical vaccine would be made from the subunit which corresponds to the head of the vaccine, for example. The “head” would be extracted from the virus to make a vaccine using that particular subunit.
Typically, a subunit vaccine is made up of components crucial to the pathogen’s infection mechanism. This is so that the human body can establish immunity to these components to immobilize components vital to the pathogens infection mechanism.
Accordingly, Novavax focuses on the spike proteins of SARS-CoV-2, which is the virus that causes COVID-19. This is because the spike proteins on SARS-CoV-2 are crucial for the virus to gain entry to human cells. Once the virus can gain entry to the cell, SARS-CoV-2 replicates and spreads, which causes COVID-19 infection.
Therefore, in the case of COVID-19, the component of interest is the viral spike protein found on the surface of the SARS-CoV-2 virus. Since NVX-CoV2373 uses a protein subunit (the virus’s spike protein), it can be classed as a protein subunit vaccine.
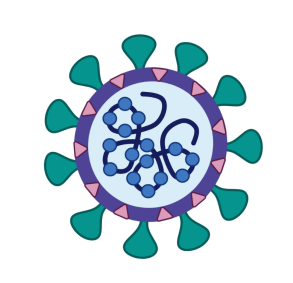
Other common subunits used in the development of these types of vaccines include viral sugars.
An advantage to using subunit vaccines of any kind is that it is capable of eliciting a very robust immune response. The strong response is due to the fact that they contain crucial parts of the pathogen to which we build immunity. However, in some cases, these vaccines could require booster shots to ensure ongoing immunity from the disease they protect against.
How Does Novavax’s COVID-19 Vaccine Work?
As we have mentioned, the component of interest in a COVID-19 vaccine is the viral spike protein found on the surface of the SARS-CoV-2 virus. This is because the SARS-CoV-2 virus is unable to cause infection without the spike proteins on its surface. Because of the spike protein’s importance in the infection mechanism, it is a promising therapeutic target.
Almost all the COVID-19 vaccines in development aim to exploit the spike protein, by either introducing information about the spike protein or the spike protein itself, to our bodies. Once our body is exposed to the spike protein, our immune systems can recognize it as an “invader” and launch an immune attack to neutralize and destroy it.
Novavax’s NVX-CoV2373 vaccine is no different from the other COVID-19 vaccines in development — it, too, exploits the viral spike protein. In the case of Novavax, the spike proteins are cultured in a laboratory using a complicated mechanism which is beyond the scope of this article due to its complexity, so we will not discuss it here. These spike proteins are then purified to remove all other molecules.
The purified spike proteins are then injected into our bodies. Once introduced, they will be recognized as “invaders” and attacked and neutralized. The introduction of spike proteins into our bodies teaches our immune cells how to attack the SARS-CoV-2 spike protein. When naturally exposed to the virus, our body will therefore have prepared its battleground and sent their best soldiers to conquer the SARS-CoV-2 virus as best as possible. Next time the body has to deal with SARS-CoV-2, these trained soldiers will be at the ready to neutralize any virus as quickly as possible to help the body avoid a COVID-19 infection.

Making the NovaVax Vaccine Requires a “Protein Factory”
To produce the spike proteins, Novavax uses a unique mechanism. This method has proven to work for the production of NanoFlu, which was fast-tracked for use against influenza. We will discuss this method using the following series of images. These infographics endeavor to simplify and explain the methods behind the NovaVax “protein factory” as best as possible.
Please note: The way in which Novavax’s vaccine is made is slightly more complicated than the other COVID-19 vaccines we have discussed so far. The following schematic, which seeks to explain the foundational concepts used, is therefore simplified to aid understanding.
The RNA coding for the SARS-CoV-2 spike protein is converted into DNA, which is added to a baculovirus. This forms a recombinant virus, containing baculovirus DNA and DNA translated from SARS-CoV-2 virus. The recombinant virus expresses the SARS-CoV-2 spike proteins when they infect cells.
As can be seen from the image above, the SARS-CoV-2 virus takes the form of an RNA molecule, a single-stranded molecule of nucleic acids. The virus is genetically engineered and chemically converted to double stranded DNA so that it can be added to another virus to facilitate mass production of the spike proteins.
The DNA engineered from the SARS-CoV-2 virus is then added to a type of virus called a baculovirus, a virus which has unique properties that make it ideal for mass producing spike proteins for use in the Novavax COVID-19 vaccine. Baculoviruses are a group of insect viruses that have a larger genome and can therefore accommodate additional “foreign” genes such as the SARS-CoV-2 virus.
The mishmash of the SARS-CoV-2 and the baculovirus is called a recombinant virus, and it contains both DNA from the baculovirus and converted DNA from the SARS-CoV-2 virus. The recombinant baculovirus can now express the SARS-CoV-2 spike protein when it infects an insect cell.
To cause infection, baculoviruses must typically be ingested by the insect. When the insect eats the recombinant virus, it can gain entry into the insect’s cells. Next, the recombinant virus we described above will take over the cellular machinery of the insect. The takeover means that the insect cells produce viruses and viral particles in bulk, which ramps up the production of the SARS-CoV-2 spike proteins that are expressed by the baculoviruses. The spike proteins can then be purified and used as a vaccine candidate.
Now that we know how the recombinant virus is made, let us look into some more detail on how Novavax manufactures NVX-CoV2373, their COVID-19 vaccine.
How is Novavax’s COVID-19 Vaccine Made?
As we have mentioned earlier, the baculoviruses must infect the insect cells so as to take over its machinery and mass produce the SARS-CoV-2 spike proteins.
For this process, Novavax uses the baculoviruses to cause infection in Sf9 insect cells. The Sf9 insect cells are a cell line that are established from the ovarian tissue of the Spodoptera frugiperda (fall armyworm moth).
Please see below for a step-by-step outline on Novavax’s manufacturing process, along with an illustration.
Please note: The following schematics aim to explain concepts of biology and are therefore simplified to aid understanding.
This illustration was created by Nidhi Parekh of The Shared Microscope using information from a published, peer-reviewed scientific paper. The purple things are the spike proteins that undergo post translational modification
- The Sf9 insect cells (these are a type of cell found in insects) are infected with the recombinant baculoviruses.
- Infection allows for viral takeover of the insect cells. The virus ensures that complex viral proteins, including the SARS-CoV-2 spike proteins, are produced. Using the cells machinery proteins are able to fold into their correct structures.
- The viral spike proteins made in Step 2 undergo further modifications using the sf9 cellular machinery. These modifications result in the production of SARS-CoV-2 spike proteins that can be transported to the surface of the cell and then be used to evoke a strong immune response.
- The spike proteins are ultimately transported to the surface of the insect cells (i.e. the cell membrane).
- Correctly folded and modified spike proteins are then extracted from the cell membrane and purified to ensure no unwanted molecules are present. These are now available to use as a major component in vaccines.
The extracted spike proteins are then inserted into “micelles” which are small artificial capsules the size of viruses. The micelles are then injected into our bodies. On injection, the micelles float freely in the human body. However, these micelles with the spike proteins do not get very far. Our immune system recognises them as “foreign” and presents them to key immune cells. This evokes the biological responses for immunity, and optimises our chances to fight off SARS-CoV-2 viruses before they can cause COVID-19.
The sf9 insect cell/baculovirus system has been widely used for the bulk production, isolation and purification of proteins. It was also used to develop vaccines against SARS-CoV which caused Middle East Respiratory Syndrome. This system was also utilised for the production of NanoFlu, a vaccine produced for older adults, which was fast-tracked for use against the seasonal influenza virus.
Clinical Trials
Although Novavax is far behind in the clinical trials compared to Moderna, Oxford/AstraZeneca and Sinovac, the vaccine shows a lot of promise. On May 25, 2020, Novavax announced that they were beginning their Phase I study of NVX-CoV2373 in Australia.
Novavax’s Phase I/II clinical trials included 130 participants with an age range of 18 to 59 years. The company is due to announce their preliminary immunogenicity and safety results for the vaccine in the first week of August. Phase II trials will begin following successful Phase I results.
Unlike the other COVID-19 vaccines in development, the NVX-CoV2373 vaccine is one of the few vaccine candidates that produces the viral spike proteins outside of the human body. The proteins are manufactured using the methods discussed above, purified, and then injected into our bodies. Other vaccine developers, such as Oxford/AstraZeneca and Moderna, are using technologies in which the genetic information relating to the SARS-CoV-2 spike protein is injected into the body, for us to then produce the spike protein.
Novavax has received $1.6 billion in funding from Operation Warp Speed to develop and distribute 100 million vaccines as early as the end of this year. This money will be put towards completing late-stage clinical trials with up to 30,000 participants and also for the establishment of large-scale manufacturing for the vaccine. They are also to receive $388 million from the Coalition for Epidemic Preparedness (CEPI) to aid in development and manufacturing of the vaccine. However this is dependent on the results of the Phase I clinical trials. Will Novavax show positive Phase I results? Keep your eyes peeled on the news for the latest on this vaccine.
This post was originally written for Fancy Comma, LLC. Check out their Twitter, Facebook, LinkedIn, or Instagram for more information, or visit their blog for more insights on effective science writing.
Related posts:
 Covid-19: Let’s Get Handsy!
Covid-19: Let’s Get Handsy!
 [Newsletter] Round-Up of COVID-19 Vaccine Updates: Week Commencing September 28, 2020
[Newsletter] Round-Up of COVID-19 Vaccine Updates: Week Commencing September 28, 2020
 [Newsletter] Round-Up of COVID-19 Vaccine Updates: Week Commencing November 09, 2020
[Newsletter] Round-Up of COVID-19 Vaccine Updates: Week Commencing November 09, 2020
 [Newsletter] Round-Up of COVID-19 Vaccine Updates: Week Commencing November 16, 2020
[Newsletter] Round-Up of COVID-19 Vaccine Updates: Week Commencing November 16, 2020
 [Newsletter] Round-Up of COVID-19 Vaccine Updates: Week Commencing January 18, 2021
[Newsletter] Round-Up of COVID-19 Vaccine Updates: Week Commencing January 18, 2021
- Written by: admin
- Posted on: August 3, 2020
- Category: COVID-19

0 comments
Vaccine Development: A Chronology – The Shared Microscope
August 3, 2020 at 5:30 pm[…] Questions on vaccine development are now very frequent, given the pressure the COVID-19 pandemic has put on healthcare systems around the world. If you want to know more about the top COVID-19 vaccines, please do have a look at some of our previous posts. We have previously written posts on the Moderna vaccine, the Oxford/AstraZeneca vaccine, the CoronaVac developed by Sinovac, and the vaccine developed by Novavax. […]
Merna Szewczyk
August 11, 2020 at 2:52 amthis just in
Why the Anti-Vax Rhetoric Prevails – The Shared Microscope
September 1, 2020 at 6:59 pm[…] vaccine, Oxford University/AstraZeneca Vaccine, the vaccine in development by Sinovac, and the Novavax vaccine) in development […]
Pfizer and BioNTech’s mRNA Vaccine Against COVID-19: How Does It Work? – The Shared Microscope
September 23, 2020 at 2:41 pm[…] like the Moderna vaccine, the Oxford University/AstraZeneca vaccine, the vaccines in development by Novavax and Sinovac, is another frontrunner in this race against the […]
news
October 18, 2020 at 11:02 amWow! You certainly covered everything in your post. I would like to read more from you. Do you run any more blogs?
[Newsletter] Round-Up of COVID-19 Vaccine Updates: Week Commencing December 07, 2020 – The Shared Microscope
December 14, 2020 at 8:37 pm[…] 9. Novavax’s NVX-CoV2373 […]
[Newsletter] Round-Up of COVID-19 Vaccine Updates: Week Commencing November 30, 2020 – The Shared Microscope
December 14, 2020 at 8:39 pm[…] 9. Novavax’s NVX-CoV2373 […]
[Newsletter] Round-Up of COVID-19 Vaccine Updates: Week Commencing November 23, 2020 – The Shared Microscope
December 14, 2020 at 8:41 pm[…] 9. Novavax’s NVX-CoV2373 […]
[Newsletter] Round-Up of COVID-19 Vaccine Updates: Week Commencing December 14, 2020 – The Shared Microscope
December 21, 2020 at 9:08 pm[…] 9. Novavax’s NVX-CoV2373 […]
[Newsletter] Round-Up of COVID-19 Vaccine Updates: Week Commencing December 21, 2020 – The Shared Microscope
December 28, 2020 at 6:04 pm[…] 9. Novavax’s NVX-CoV2373 […]
[Newsletter] Round-Up of COVID-19 Vaccine Updates: Week Commencing December 28, 2020 – The Shared Microscope
January 4, 2021 at 7:49 pm[…] 9. Novavax’s NVX-CoV2373 […]
[Newsletter] Round-Up of COVID-19 Vaccine Updates: Week Commencing January 04, 2020 – The Shared Microscope
January 11, 2021 at 6:55 pm[…] 9. Novavax’s NVX-CoV2373 […]
[Newsletter] Round-Up of COVID-19 Vaccine Updates: Week Commencing January 11, 2021 – The Shared Microscope
January 18, 2021 at 4:22 pm[…] 9. Novavax’s NVX-CoV2373 […]
[Newsletter] Round-Up of COVID-19 Vaccine Updates: Week Commencing January 18, 2021 – The Shared Microscope
January 25, 2021 at 8:01 pm[…] 9. Novavax’s NVX-CoV2373 […]
[Newsletter] Round-Up of COVID-19 Vaccine Updates: Week Commencing January 25, 2021 – The Shared Microscope
February 1, 2021 at 5:49 pm[…] 9. Novavax’s NVX-CoV2373 […]
[Newsletter] Round-Up of COVID-19 Vaccine Updates: Week Commencing February 01, 2021 – The Shared Microscope
February 8, 2021 at 8:57 pm[…] 9. Novavax’s NVX-CoV2373 […]
[Newsletter] Round-Up of COVID-19 Vaccine Updates: Week Commencing February 08, 2021 – The Shared Microscope
February 15, 2021 at 7:32 pm[…] 9. Novavax’s NVX-CoV2373 […]
[Newsletter] Round-Up of COVID-19 Vaccine Updates: Week Commencing January 04, 2021 – The Shared Microscope
August 1, 2021 at 10:46 pm[…] 9. Novavax’s NVX-CoV2373 […]